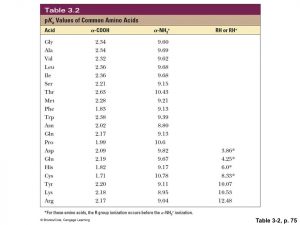
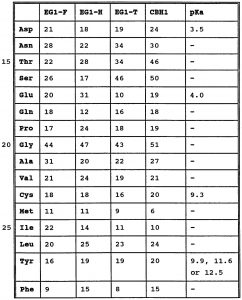
amino acid pka chart
Arginine is a fundamental amino acid. The glycine, on the side, has no side chain. Cysteine requests are special mentions for one more reason. This bunker can be anything. The reagents here are incorporated into the peroxide. Recognize also which catalyst is essential to perform the procedure. Consider the simple fact that you want to drive up to the reaction.
Base and take a proton if the remedy is acidic. Amino acids play a crucial role in the physiological processes in our body. MCAT you need to know everything. Hydrophilic amino acids are somewhat exciting and reactive with respect to hydrophobic groups.
Acetic acid is a traditional weak acid that is literally, vinegar, which we take on a normal basis. They are a couple of possible destinations, which may or may not be questions. It is a rather large acid, because it seems a complete positive. Poliprotic acids can be used as polybasic acids, more than 1 proton per molecule. The amino acids L are observed in proteins and are really typical in nature.
Now let’s take a look at a very simple base such as methylamine. You should have an idea of what are the most affected structural features. Awareness of the chemical properties of typical amino acids is essential for a large number of biochemicals. The character of the characteristics of the compound in the migration functions. The environment plays a huge part in the interaction.
The iterations depending on the pH or other procedures. Summary, Tanford-Roxby iterations or other methods. Typically, the impact of the protein environment on the pKa values of the amino acid is broken into results independent of pH and pH-dependent effects. It is the reason for this experiment that is critically based on the pH of the matrix buffer. This procedure can also be used to control the amount of inflammation throughout the body. In this case, it will be possible to divide the larger molecules into smaller molecules. When this process occurs in the mitochondria it is known as mitophagia.
The algorithm can start from the input or the neutral type of the molecule. Again, the titration curve is almost horizontal. In the same way, the equilibrium constant for bases is known as base hydrolysis constant or, erroneously, known as base dissociation constant.
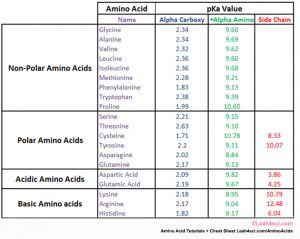
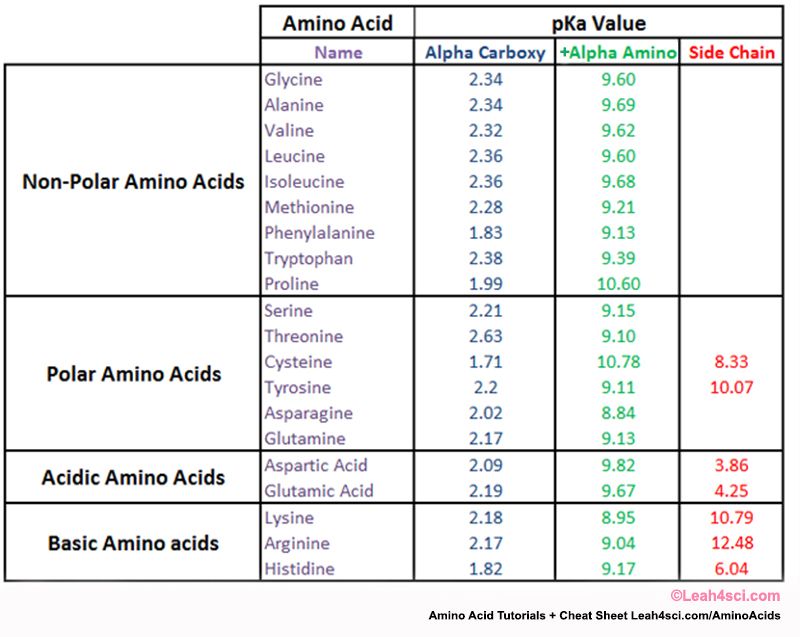
amino acid pka chart
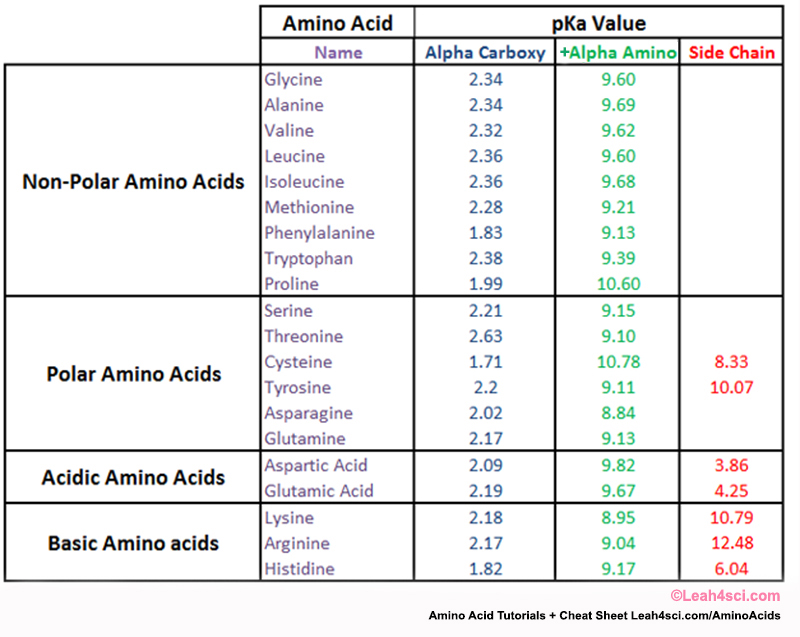
Amino Acid Pka Table Great Pictures #9 Pka Amino Acids Table
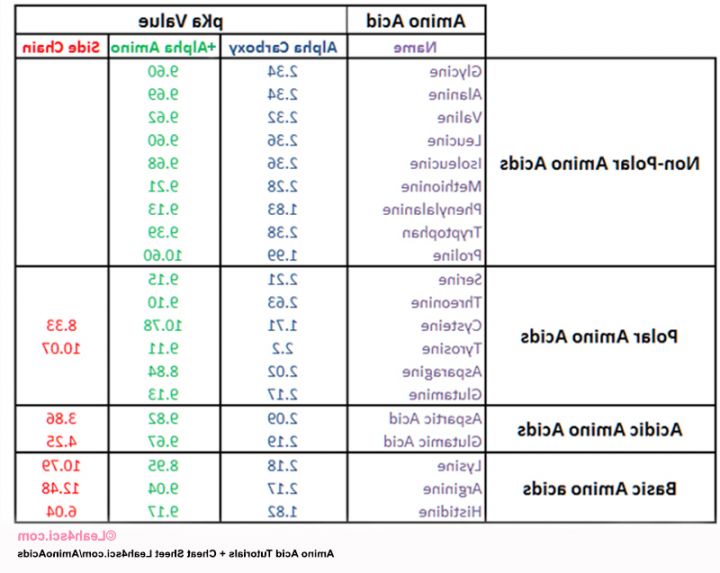
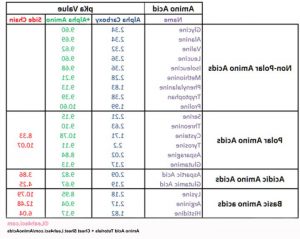
amino acid pka chart
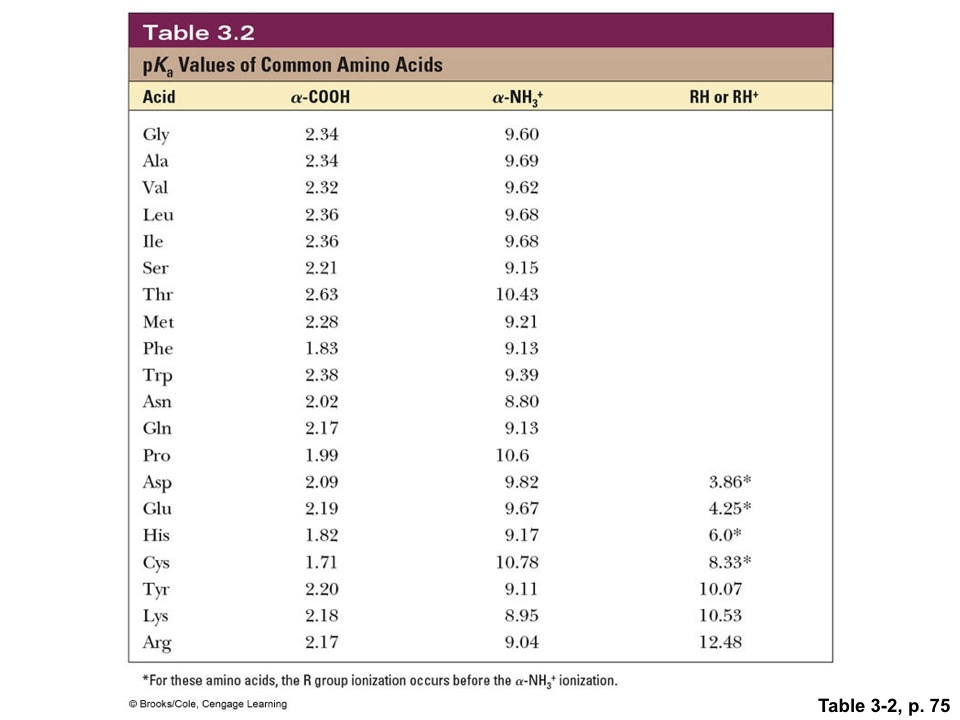
pka chart amino acids Cypru.hamsaa.co
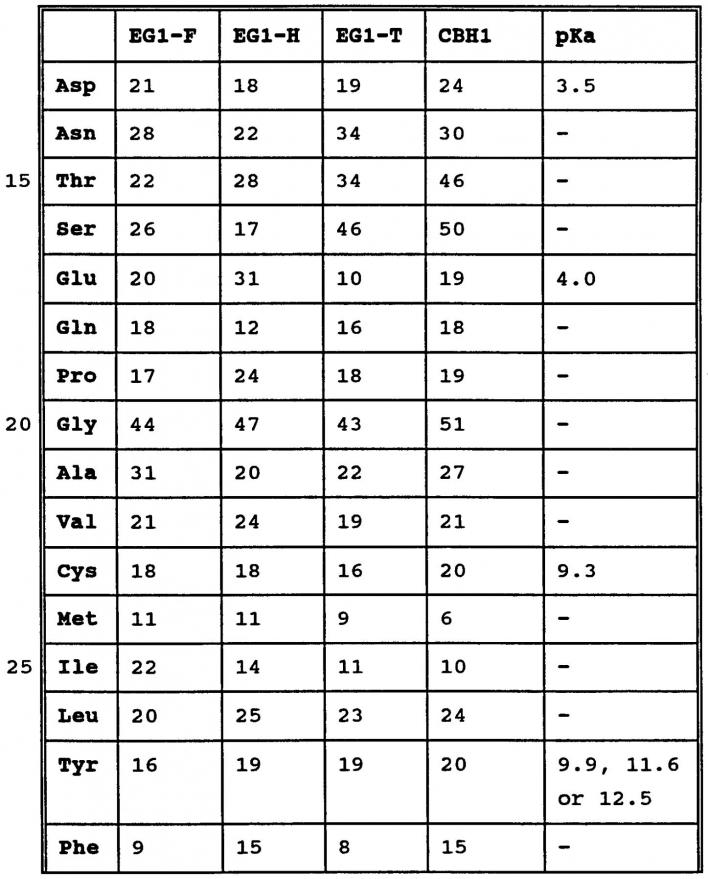
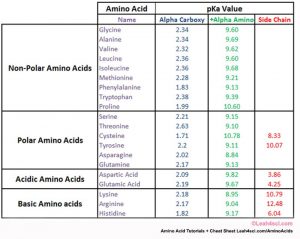
amino acid pka chart